 
In this case number of moles of reactants are greater than the products and the product is in solid-state so the change in entropy of a system is negative. Similarly, if there is a greater number of moles in reactants than products there will be a negative change in entropy. So if reactants are in the gas phase and products are in the solid phase then there will be a negative change in entropy. Change in states of reactants and productsĪs gases have high entropy and solids have low entropy.There are two rules to identify the change in entropy. In a chemical reaction, by comparing the entropies of reactants and products we can determine whether the entropy increase or decreases. Bromine has high entropy because it tends to spread out. For example, diamond has lower entropy than graphite because diamond atoms are arranged in regular order. So entropy change of system will be negative.Ĭarbon monoxide CO has less entropy than carbon dioxide CO 2 because simpler substances with fewer atoms have lower entropy values than more complex substances with a greater number of atoms.įor similar types of substances, harder substances have a lower entropy value. As the randomness of the system decrease so the change of entropy will be negative.ĭuring the condensation process i.e gas is converted into a liquid system moves from a less ordered state to a more ordered state due to a decrease in volume and temperature. This is because that particles of liquids are more disordered than solid particles. When liquid is converted into solid i.e freezing process the transition takes place from high entropy to low entropy. Concepts Berg Examples of negative change in Entropy.Entropy changes for exothermic and endothermic reactions.Instead, we can only determine the change in entropy.Īt a given temperature heat is transferred reversibly in and out of the system. We can never determine the absolute value of entropy. According to the 2nd law of thermodynamics, the entropy of the universe for a spontaneous process is always increasing because everything in the universe tends towards a more disordered state spontaneously. The randomness of the system increases, its entropy increases. They are more disordered so they have large entropy. In the case of gases, the particles are far away from each other. The case of liquids is intermediate between solids and liquids. 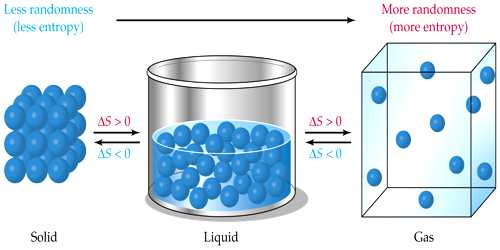
In the case of solids, the particles are very close to each other because they are arranged in regular order, so solids have less entropy. More randomness in a system corresponds to more entropy. It is a measure of the randomness or disorder of a system. Entropy is a measure of the dispersal of energy at a specific temperature from the system to the surrounding or otherwise. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
